Blog
Blog – July 02, 2025
Target Malaria Uganda and Burkina Faso teams share stakeholder engagement experiences

Blog – June 30, 2025
Day of the African Woman 2025

Blog – June 27, 2025
Partnering with Diptera.ai to build the tech capacity of African research institutes to use the technologies of tomorrow

Blog – June 10, 2025
Communicating our science at the Great Exhibition Road Festival

Blog – June 02, 2025
Building community bonds through community engagement in Koome sub-county

Blog – May 26, 2025
Africa Day 2025: We are joining the #CorrectTheMap movement

Blog – May 19, 2025
“It’s time to change the story”: Target Malaria scientists featured in the new Change the Story campaign

Blog – May 12, 2025
African Parliamentarians Unite to Fight Malaria: COPEMA launches in Accra, Ghana

Blog – May 02, 2025
Target Malaria Ghana issues a glossary on Anopheles gambiae and organisms sharing its habitat in Abutia Amegame and Mafi Agorve in the Volta Region of Ghana

Blog – April 25, 2025
Together we can end malaria

Blog – April 23, 2025
Celebrating World Malaria Day 2025

Blog – April 15, 2025
Bridging science and community in Kalangala

Blog – April 10, 2025
Malaria, machine learning, and the power of collaboration, a discussion with the Young Scientists’ Network

Blog – April 09, 2025
Celebrating private sector partners’ transformative impact on the fight against HIV, TB and malaria

Blog – April 02, 2025
European leadership in global health advancement: A keystone to the reach for global health equity

Blog – March 24, 2025
AUDA-NEPAD launches the 2nd edition APET report on gene drives for malaria control and elimination

Blog – March 18, 2025
RBM Vector Control Working Group 2025 meeting in Cotonou

Blog – March 11, 2025
A maternal health perspective on malaria

Blog – February 19, 2025
Building new bridges between Target Malaria Uganda and Target Malaria Burkina Faso

Blog – February 11, 2025
Celebrating the women who advance science

Blog – January 29, 2025
Target Malaria’s 2024 Highlights

Blog – January 28, 2025
Malaria: A call for action

Blog – December 18, 2024
Reflecting on my journey in Malaria Advocacy with Target Malaria’s Young Scientists Network

Blog – November 21, 2024
Predicting the potential of gene drives in mosquitoes to control malaria in West Africa with mathematical models

Blog – November 20, 2024
Dr. Abdoul Azize Millogo: Pioneering GIS Solutions for Vector-Borne Disease Control in Burkina Faso

Blog – November 18, 2024
Mapping Africa-driven innovation to fight malaria

Blog – November 15, 2024
COP 16 highlights: A new path for peace with nature

Blog – November 14, 2024
“Voices from the field”: the story of an encounter

Blog – November 07, 2024
What do genetically modified mosquitoes look like?

Blog – October 31, 2024
Science symposiums in Burkina Faso

Blog – October 25, 2024
Embracing innovation in science reporting: My journey and reflections

Young Scientists – October 23, 2024
Three Questions to a Scientist: Mahamadi Kientega, medical entomologist in Burkina Faso

Blog – October 17, 2024
New Scientist Live 2024

Blog – October 10, 2024
Understanding the mosquito: Target Malaria Ghana’s mesocosm study

Blog – October 03, 2024
How to dissect a mosquito

Blog – September 11, 2024
Can the Anopheles gambiae mosquito pollinate? A scientific exploration

Blog – August 19, 2024
World Mosquito Day 2024

Blog – August 15, 2024
Mosquito-borne diseases and their vectors

Blog – August 07, 2024
Anopheles, Aedes or Culex: how to tell mosquito species apart?

Blog – July 26, 2024
Junior high school students visit the mosquito ecology research facility in Ghana
Blog – July 18, 2024
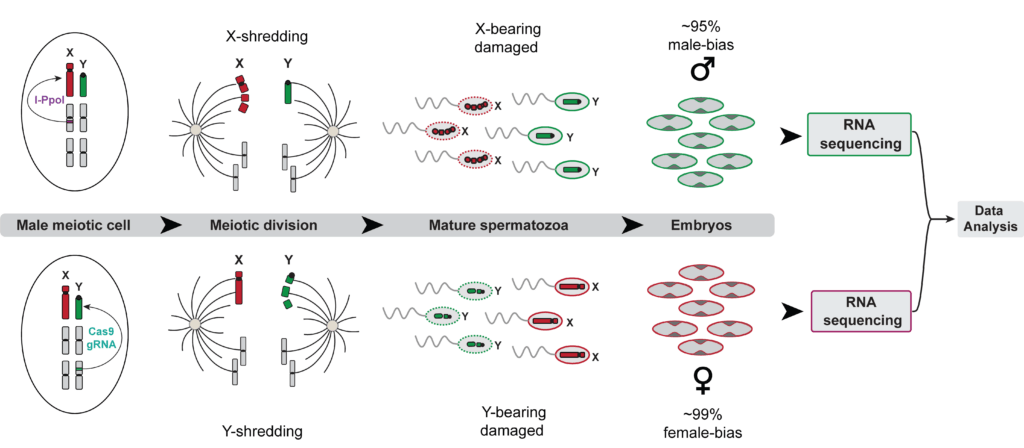
Y chromosome shredding in Anopheles gambiae: a new genetic sexing strain reveals novel insight into the biology of synthetic sex ratio distorters

Blog – July 12, 2024
What would a malaria-free future look like?

Blog – June 19, 2024
The Great Exhibition Road Festival 2024

Blog – June 14, 2024
Target Malaria team participates in biodiversity negotiations (SBSTTA-26)

Blog – May 30, 2024
Joining the Malaria Technical Expert Collective

Blog – May 24, 2024
Designs on field testing of gene drive for malaria control

Blog – May 24, 2024
Africa Day 2024
Blog – May 16, 2024
Where gene drive fits into WHO’s new Global Malaria Programme operational strategy

Blog – May 09, 2024
What is the role of a Stakeholder Engagement and Communications Officer in Ghana?

Blog – May 03, 2024
Reflecting on the RBM Vector Control Working Group and Multi-sectoral Working Group Meetings

Blog – April 25, 2024
Celebrating World Malaria Day 2024

Blog – April 22, 2024
World Malaria Day: Accelerating the fight against malaria for a more equitable world

Blog – April 18, 2024
Approaching World Malaria Day 2024

Blog – April 01, 2024
What is the job of a stakeholder engagement expert?

Blog – March 14, 2024
Yaoundé Declaration from the Malaria Ministerial Meeting

Blog – February 26, 2024
World NTD Day 2024

Blog – February 19, 2024
Imperial Lates: Mozzie mating

Blog – February 12, 2024
Our Work in Ghana
Blog – January 25, 2024
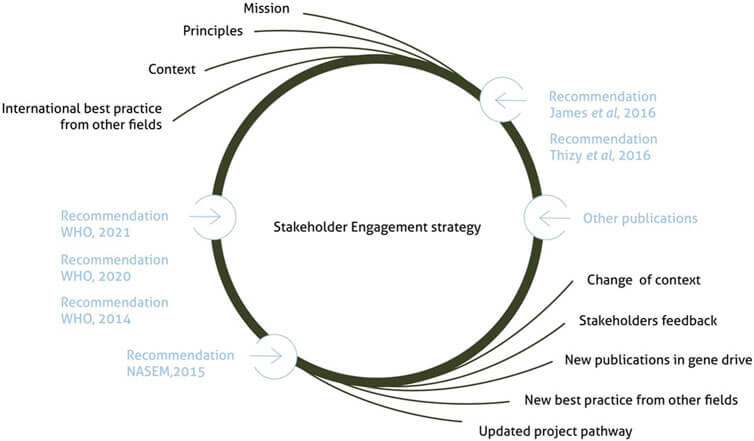
Stakeholder engagement in the development of genetically modified mosquitoes for malaria control in West Africa: lessons learned from 10 years of Target Malaria’s work in Mali

Blog – January 11, 2024
Target Malaria’s stakeholder engagement approach

Blog – December 21, 2023
Communicating creatively about genetically modified mosquitoes

Blog – December 15, 2023
Watching insecticide resistance evolve over time in Burkina Faso

Blog – December 12, 2023
Gene drive technology unlocks innovative potential solutions at the intersection of climate change and public health

Blog – December 01, 2023
World Malaria Report 2023: a call for concerted action to address growing threats
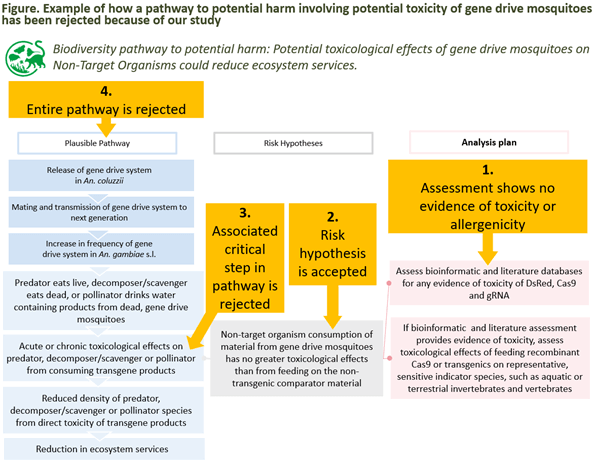
Blog – November 14, 2023
Assessment of the potential for toxicity and allergenicity from our gene drive mosquitoes

Blog – November 08, 2023
World Quality Week: What it means to be a quality assurance officer at Target Malaria

Blog – October 31, 2023
Inclusive Health Systems: The Key to Addressing Global Health Challenges

Blog – October 26, 2023
Capturing the Journey of Target Malaria Ghana: A Visual Narrative

News – October 11, 2023
Highlights from the PAMCA Conference in Addis Ababa

Blog – September 01, 2023
A dual perspective on targeting malaria vectors

Blog – August 25, 2023
Climate change and malaria mosquitoes: A looming public health crisis

Blog – August 18, 2023
World Mosquito Day: Target Malaria’s 2023 #MosquitoCountdown

Blog – August 11, 2023
Microinjection: Transformation from wild to genetically modified mosquito

Blog – August 04, 2023
Why World Mosquito Day?
Blog – July 14, 2023
Empowering communities in the fight against malaria: Reflections from the Gordon Research Conference

Blog – July 07, 2023
Celebrating five years of Zero Malaria Starts with Me

Blog – June 30, 2023
A photographic look at the future based on a rich past

Blog – June 22, 2023
Communicating science creatively at the Great Exhibition Road Festival

Blog – June 15, 2023
My role as a malaria ambassador

Blog – June 02, 2023
How Target Malaria uses theatre to communicate with communities
Blog – May 26, 2023
What does it mean to be an entomologist?

Blog – May 19, 2023
Our work at the CDC Foundation for Target Malaria

Blog – May 04, 2023
Malaria researchers celebrated as new “rock n roll stars” this World Malaria Day

Blog – April 25, 2023
Meet the team innovating to End Malaria

Blog – April 25, 2023
Celebrating World Malaria Day 2023

Blog – April 18, 2023
Time to Deliver Zero Malaria

Blog – March 24, 2023
How do communities the project engages with make informed decisions about our work?
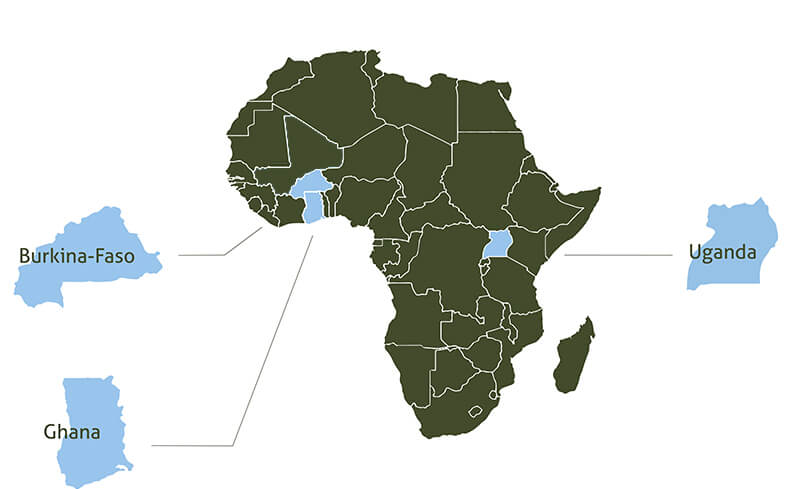
Blog – March 17, 2023
Reintroducing where we work

Blog – March 08, 2023
International Women’s Day: It’s time to #EmbraceEquity

Blog – March 03, 2023
What do you need to know about malaria?

Blog – February 17, 2023
The RBM Partnership to End Malaria’s Vector Control Working Group meeting

Blog – January 27, 2023
Reflections on 2022
Blog – January 11, 2023
What role do malaria mosquitoes play in their ecosystem?

Blog – December 21, 2022
Meet Anopheles stephensi, the mosquito threatening to derail malaria prevention efforts in Africa
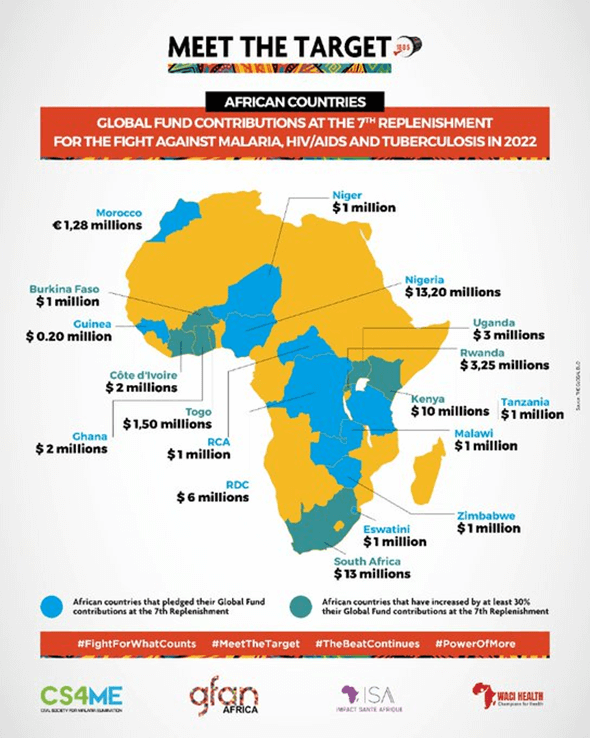
Blog – December 14, 2022
African leaders prioritise health at the Global Fund replenishment

Blog – December 02, 2022
East African policy dialogue on research of genetically modified mosquitoes for malaria control and elimination

Blog – November 30, 2022
How the climate crisis could affect malaria transmission
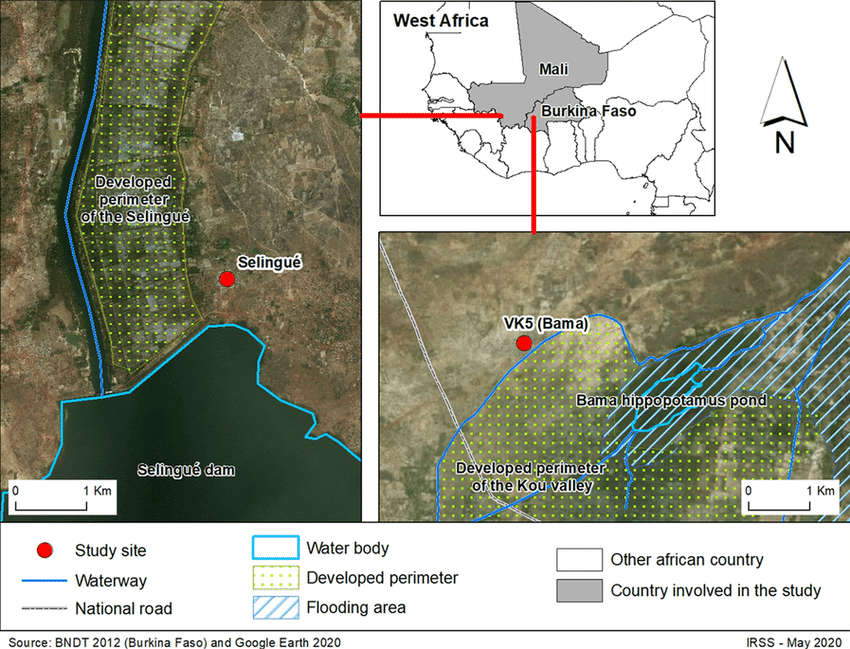
Blog – November 16, 2022
How Earth Observation Data is used by Target Malaria

Blog – October 19, 2022
Innovating to end malaria, from science to stakeholder engagement
Blog – October 14, 2022
Reflections on the 8th Annual PAMCA Conference

Blog – October 05, 2022
Antimalaria drug resistance: an endless run

Blog – September 07, 2022
What are the geographical factors that affect malaria transmission?

Blog – August 24, 2022
Operationalizing stakeholder engagement guidance for gene drive research

Blog – August 03, 2022
Overcoming genome sequencing gaps to accurately characterise a genetically modified mosquito strain

Blog – July 27, 2022
What do we mean by “Target Organism” in Target Malaria’s gene drive research?

Blog – July 15, 2022
A multi-disciplinary approach for building a common understanding of genetic engineering for malaria control in Burkina Faso

Blog – July 08, 2022
Introducing the African Gene Drive for Vector Control Network

Blog – June 15, 2022
How to advance environmental risk assessment of gene drive for malaria when it’s never been done before

Blog – June 09, 2022
Celebrating a golden age of African malaria research

Blog – May 17, 2022
Updating the terminology to describe our genetically modified mosquitoes

Blog – May 11, 2022
Importation of the non gene drive genetically modified male bias mosquito strain into Burkina Faso

Blog – May 09, 2022
Meet the Target Malaria scientists harnessing innovation to end malaria

Blog – May 05, 2022
Preparing Africa’s first facility for the study of transgenic mosquitoes

Blog – March 30, 2022
Translating scientific concepts into local African languages

Blog – March 17, 2022
Meet the people at Target Malaria helping #BreakTheBias this International Women’s Day

Blog – March 01, 2022
Communicating with communities through microprogrammes

Blog – February 22, 2022
My appointment as a Fellow to the African Academy of Sciences (AAS)
Blog – February 11, 2022
Prix d’excellence de la Pan-African Mosquito Control Association pour les femmes dans le domaine du contrôle vectoriel

Blog – February 11, 2022
The Pan-African Mosquito Association’s Women in Vector Control Excellence Awards

Blog – February 10, 2022
Entomological results from the first release of non gene drive genetically modified sterile male mosquitoes in Africa

Blog – February 08, 2022
Ethical principles guiding Target Malaria’s engagement strategy

Blog – January 13, 2022
Expedition to collect mosquitoes in Uganda

Blog – January 11, 2022
Reflections on 2021

Blog – December 02, 2021
Arthropod Containment Guidelines updated with addendum on gene drive
Blog – November 24, 2021
Understanding the malaria vector: our entomological work in Burkina Faso

Blog – November 09, 2021
Two years of laboratory studies on the non gene drive genetically modified sterile male mosquitoes concluded successfully in Mali

Blog – November 05, 2021
Providing a policy framework for responsible gene drive research

Blog – October 27, 2021
Developing a stakeholder engagement roadmap for novel genetic approaches for vector control

Blog – October 12, 2021
WHO recommends malaria vaccine for broad-scale use in Africa

Blog – September 22, 2021
Investigating the potential ecological consequences of gene drive approaches to vector control

Blog – September 06, 2021
Gene drive mosquitoes successfully control mosquito populations in large cages

Blog – July 29, 2021
World Health Assembly resolution prioritises research and innovation for malaria control

Blog – July 08, 2021
Analysing off-target effects in CRISPR-based gene drive mosquitoes

Blog – June 30, 2021
Talking public health, biodiversity and development at the European Development Days
Blog – June 09, 2021
Laying the groundwork for a community agreement framework for gene drive research in Africa

Blog – May 25, 2021
Building the Africa we want through science and innovation

Blog – May 19, 2021
Disparities in malaria risk and resilience amongst rural populations in western Burkina Faso

Blog – April 15, 2021
Research must be a collaborative effort

Blog – March 31, 2021
Results from months of monitoring following the first release of non gene drive genetically modified mosquitoes in Africa

Blog – March 29, 2021
Investigating potential harms from the use of gene drive mosquitoes to fight malaria

Blog – March 24, 2021
Fighting malaria in the field through entomology

Blog – March 12, 2021
Joining the GFAN 2021-22 Speakers Bureau

Blog – February 25, 2021
Draw the line against malaria!

Blog – February 10, 2021
Using DNA barcodes in the fight against malaria

Blog – February 03, 2021
Making Target Malaria’s science accessible to kids

Blog – January 27, 2021
Co-Developing a common glossary with stakeholders

Blog – January 18, 2021
2021: Progress during challenging times

Blog – January 11, 2021
Spanish TV Science Series visits the Crisanti Lab

Blog – December 03, 2020
Malaria must die so millions can live

Blog – November 30, 2020
World Malaria Report: We saved millions, but we need to do more

Blog – November 24, 2020
Reshaping Nature through Gene Drives – A Podcast

Blog – November 17, 2020
Why does the development of innovative technologies require a strong community engagement in rural Africa?

Blog – November 10, 2020
Malaria’s growing financial dilemma

Blog – November 04, 2020
WHO supports research on genetically modified mosquitoes for malaria control

Blog – October 22, 2020
Why eradicating Malaria in Africa requires a specific approach and new tools?

Blog – October 15, 2020
Investment in new vector control tools recommended if we are to avoid malaria resurgence, new study indicates

Blog – October 06, 2020
Target Malaria Uganda Starting Work on Colour-Variant Mosquitoes

Blog – August 25, 2020
Understanding the population dynamics of a self-limiting male bias strain in malaria mosquitoes

Blog – August 20, 2020
Virtual tours of Target Malaria’s insectaries to celebrate World Mosquito Day

Blog – August 12, 2020
International Youth Day: Youth Engagement for Malaria Elimination

Blog – August 11, 2020
Malaria, tuberculosis, and HIV : how can the impact of COVID-19 be minimised
Blog – August 03, 2020
What is COVID-19 teaching us about sustainable development?
Blog – July 24, 2020
Bridging the investment gap in the fight against malaria
Blog – July 15, 2020
Global fund alerts to the effects of COVID-19 pandemic on HIV, tuberculosis and malaria
Blog – June 30, 2020
Happy 15th Anniversary, U.S. President’s Malaria Initiative!
Blog – June 29, 2020
Crisanti lab on lockdown
Blog – June 22, 2020
Community engagement: Why collaborate with an entomological study?
Blog – May 25, 2020
Target Malaria Ghana: Unlocking DNA and Ecosystem Knowledge
Blog – May 11, 2020
Target Malaria scientists create a genetically modified mosquito to produce male-only progeny that effectively reduces the population of malaria mosquitoes in the lab at Imperial College

Blog – April 24, 2020
World Malaria Day 2020: Our Champions
Blog – April 24, 2020
World Malaria Day 2020: Our Countries

Blog – April 07, 2020
Health workers, thank you for making the difference

Blog – March 27, 2020
COVID-19: We are here for you

Blog – March 20, 2020
Target Malaria Burkina Faso receives the prestigious 2019 Newcomb Cleveland Prize at the AAAS

Blog – March 09, 2020
My Interview on Target Malaria in Italian

Blog – February 20, 2020
The ecology unit at Polo GGB: rearing modified mosquitoes in large cages

Blog – January 08, 2020
Dr Mamadou Coulibaly

Blog – August 09, 2019
CRISPRcon 2019

Blog – July 01, 2019
Target Malaria proceeded with a small-scale release of genetically modified sterile male mosquitoes in Bana, a village in Burkina Faso

Blog – March 29, 2019
What is modelling and why is it important in our research?
Blog – February 01, 2019
2018 – Key Achievements
Blog – December 04, 2018
Feedback on the Convention on Biological Diversity
Blog – November 23, 2018
Burkina Faso is getting ready for its next stage of research – sterile male mosquito release

Blog – November 16, 2018
Open Letter: “Research on gene drive technology can benefit conservation and public health”
Blog – October 02, 2018
Meet our team: Dr Tony Nolan
Blog – July 30, 2018
‘If you succeed, what about the other animals?’

Blog – July 06, 2018
Our stakeholder engagement approach in Uganda

Blog – June 04, 2018
Meet our team: Professor Sir Charles Godfray

Blog – April 25, 2018
World Malaria Day, a time to recommit.
Blog – April 19, 2018
Commonwealth Malaria Summit 2018 #readytobeatmalaria
Blog – March 21, 2018
How the stakeholder engagement teams of Target Malaria work together

Blog – February 21, 2018
Meet our team: Krystal Birungi, Field Entomology Coordinator at the Uganda Virus Research Institute
Blog – December 07, 2017
Imperial academic puts community at the heart of malaria elimination project
Blog – December 07, 2017
Channel 4, Fight against malaria has stalled, World Health Organisation says
Blog – December 07, 2017
Nature, Genetic engineering: On the road to efficient gene drives
Blog – December 07, 2017
Can Scientists Convince the Public to Accept CRISPR and Gene Drives?
Blog – December 07, 2017
Outwitting nature’s greatest killer
Blog – November 21, 2017
How Homing Endonucleases can be used to cut DNA
Blog – October 18, 2017
A day in Bobo-Dioulasso
Blog – September 13, 2017
The long journey of mosquitoes – Insight from London

Blog – August 08, 2017
New Partnership for African Development workshop
Blog – July 05, 2017
Terni Open Days

Blog – June 16, 2017
Dr. Abdoulaye Diabaté give us an insight into Target Malaria’s team work in Burkina Faso
Blog – May 10, 2017
SciDev, GM drive against malaria: Treading a fine line, April 2017
Blog – May 10, 2017
Stat News, In a remote West African village, a revolutionary genetic experiment is on its way — if residents agree to it, March 2017
Blog – May 10, 2017
Wired, Illustrated experiment: modifying mosquitoes to save millions of lives, March 2017
Blog – May 10, 2017
The Scientist , Using gene drive to limit the spread of malaria , January 2017
Blog – May 10, 2017
The New Yorker, Rewriting the code of life, January 2017
Blog – May 10, 2017
Nature, ‘Gene drive’ moratorium shot down at UN biodiversity meeting, January 2017
Blog – April 25, 2017
The pressing need for new vector control tools
Blog – April 03, 2017
Target Malaria in Uganda is shaping up
Blog – February 20, 2017
Our team development
Blog – February 07, 2017
Meet Our Team: Dr. Lea Toe, Stakeholder Engagement Leader
Blog – January 18, 2017
Key Achievements of 2016
Blog – December 05, 2016
Open Letter on Gene Drive Technology

Blog – November 17, 2016
Target Malaria Presents at Grand Challenges
Blog – October 25, 2016
Gene drives: The good, the bad, and the hype

